1. Background
Digital mammography (DM) is one of the most common and effective radiological methods for breast cancer screening and detection (1). Mammographic sensitivity and specificity may be decreased by factors, such as dense breast tissue, which may lead to a low positive predictive value (PPV) and low sensitivity (2). Dense fibroglandular structures in a normal breast tissue can suggest lesions, although a dense fibroglandular tissue can cause false negativity by superimposing on the lesion margins. To improve cancer diagnosis in patients with dense breast parenchyma, in breast imaging-reporting and data system (BIRADS) 0 lesions, in indeterminate BI-RADS categories 3 and 4 lesions, additional imaging techniques are needed.
Ultrasonography (US) is an adjuvant modality, especially for the management of indeterminate breast lesions with underlying dense breast parenchyma, and is complementary to mammography (3). Besides, US is an effective method for the examination of lesions in dense breasts, as it is less affected by tissue overlapping (4). Digital breast tomosynthesis (DBT) is a modality, eliminating the tissue overlap and enabling radiologists to see through the structural noise of a normal breast tissue (5). Overall, DBT is a modified version of DM, which can combine three-dimensional and cross-sectional views with other algorithms to produce an image (6).
2. Objectives
The current study aimed to evaluate the contribution of US and DBT to DM in patients with dense and non-dense breast parenchyma and patients with BI-RADS 0 lesions and BI-RADS 3 - 4 lesions. Whether US and DBT can upgrade or downgrade the BI-RADS category of an uncertain lesion detected on DM was also investigated. Moreover, the effect of breast density on the US and DM contribution was examined.
3. Patients and Methods
In this study, a total of 120 patients, who underwent DM and DBT between 2017 and 2018, were examined. According to DM, patients with BI-RADS 0, 3, and 4 lesions were included. On the other hand, patients with typical benign findings (BI-RADS 1 and 2 lesions) and typical malignant findings (BI-RADS 5 lesions) were excluded from the study. Also, patients with a history of surgery or chemotherapy were eliminated from the study.
DM followed by DBT was performed for the patients, and BI-RADS categorization system was applied based on the results. Subsequently, the patients were evaluated sonographically using US, and the BI-RADS categorization system was employed. Finally, 46 patients, who were classified as BI-RADS categories 0, 3, and 4 on DM, according to DBT and US findings.
This study was conducted based on the ethical standards outlined in the Declaration of Helsinki of the World Medical Association. Ethical approval was obtained from a regional ethics committee.
3.1. Image Acquisition
All patients underwent DM using a commercially available system (Mammomat Inspiration, Siemens, Erlangen, Germany); a high milliamperes was among the features of this system. An anode system, made of tungsten and rhodium filters, could extend up to 15 degrees and produce images with a slice thickness of 1 mm. Moreover, a Giotto Breast Tomosynthesis system (IMS, Bologna, Italy) was used for DBT. DBT imaging was carried out for all patients. The imaging data of DM and DBT were stored in our local database. Subsequently, the patients were examined sonographically, using a commercial Logiq 7 USG device (General Electrics, USA) with a 10-14 MHz linear probe. For the USG examination, the patients were asked to lie in a supine position, and a breast scan was performed to find lesions in both radial and axial planes.
3.2. Image Analysis
The images and the patients were examined by two radiologists (with four and 20 years of experience in breast conditions, respectively). DBT, US and US were performed on the patients, respectively. The researchers were blinded to the histopathological and clinical data while evaluating the images. First, the breast density was categorized as A, B, C, and D on DM; this categorization was based on to the 2013 American College of Radiology (ACR) breast atlas. Besides, three different BI-RADS classifications (one for DM, one for DBT, and one for US) were described for each modality; A and B represented non-dense fatty breast patterns, while C and D represented dense breast patterns.
The lesion site, focal asymmetry with microcalcifications, and other suspicious findings were first evaluated on DM and then on DBT. Craniocaudal (CC) and mediolateral oblique (MLO) images were acquired for breast lesions on DM. DBT images were interpreted in the light of the information provided by the DM. DBT images were evaluated either in CC or MLO views. The two radiologists reached a consensus and made a final decision. Besides, a core needle biopsy was performed, and the pathological findings were considered as the reference standard.
The patients who did not undergo biopsy were identified as multimodality benign. A multimodality benign patient was benign and showed no progression during two years of follow-ups.
3.3. Statistical Analysis
All statistical analyses were performed in R software version 3.6.0 (www.r-project.com). Descriptive statistics for the patients’ demographic and lesion characteristics are presented as mean ± standard deviation (SD), count (n), and percentage (%). Sensitivity, specificity, PPV, and negative predictive value (NPV) were calculated at 95% confidence intervals (CIs) to compare the diagnostic performance of screening tests alone and in combination. Moreover, the sensitivity and specificity of the tests were compared using the McNemar’s test. The PPV and NPV were compared according to the weighted generalized score statistic. On the other hand, the diagnostic performance of DBT and US for the detection of malignant lesions was compared based on a receiver operating characteristic (ROC) curve analysis. Besides, the accuracy of DBT and US was examined separately, and then, the accuracy of DM combined with US or DBT was evaluated. Diagnostic accuracy was also calculated for the combined techniques. A true positive or false positive result was defined as at least one positive or negative result in the three imaging studies, while a true negative result was defined as three compatible negative imaging results. Moreover, the effects of breast density on the diagnostic performance of modalities were evaluated by the ROC curve analysis. P-value less than 0.05 was considered statistically significant.
4. Results
The age range of the patients was 35-70 years, with a mean age of 51.7 years. Most of the lesions (56%) were located in the right breast. Also, 69% of the lesions were located in dense breast parenchyma. Based on the findings, 69% of the lesions were histopathologically confirmed as benign; most of them were benign based on multiple modalities. Invasive ductal carcinoma accounted for the majority of malignant lesions (78%) (Table 1). The BI-RADS categories and benign or malignant presentations of lesions based on DM + DBT and US are summarized in Table 2.
| Characteristics | Patients (n = 46) |
|---|---|
| Age, mean ± SD (min-max) | 51.7 ± 7.65 (35-70) |
| Side of lesion, No. (%) | |
| Right breast | 26 (56.5) |
| Left breast | 20 (43.5) |
| Breast density, No. (%) | |
| A | 1 (2.2) |
| B | 13 (28.3) |
| C | 28 (60.9) |
| D | 4 (8.7) |
| Diagnosis of lesion | |
| Benign | 32 (69.6) |
| Multimodal benign | 17 (53.1) |
| Usual ductal hyperplasia | 3 (9.4) |
| Fibrocystic changes | 3 (9.4) |
| Fibrosis | 2 (6.3) |
| Duct ectasia | 1 (3.1) |
| Fibroadenoma | 1 (3.1) |
| Fibrosis adenosis | 1 (3.1) |
| Granulomatous mastitis | 1 (3.1) |
| Hamartoma | 1 (3.1) |
| Intramammary lymph nodes | 1 (3.1) |
| Sclerosing adenosis | 1 (3.1) |
| Malignant | 14 (30.4) |
| Invasive ductal carcinoma | 11 (78.6) |
| Ductal carcinoma in situ | 1 (7.1) |
| DCIS + IDC | 1 (7.1) |
| Invasive lobular carcinoma | 1 (7.1) |
Abbreviations: SD, standard deviation; DCIS, ductal carcinoma in situ; IDC, invasive ductal carcinoma.
a Data are described as mean ± standard deviation (range: min-max) or number (n) and percentage (%).
| DM | DBT | US | ||||
|---|---|---|---|---|---|---|
| Malignant | Benign | Malignant | Benign | Malignant | Benign | |
| BI-RADS 0 | 5 (10.9) | 2 (4.3) | ||||
| BI-RADS 1 | 0 | 10 (21.7) | 0 | 10 (21.7) | ||
| BI-RADS 2 | 1 (2.2) | 11 (23.9) | 0 | 6 (13) | ||
| BI-RADS 3 | 3 (6.5) | 26 (56.5) | 0 | 1 (2.2) | 0 | 4 (8.7) |
| BI-RADS 4 | 6 (13) | 4 (8.7) | 3 (6.5) | 6 (13) | 4 (8.7) | 12 (26.1) |
| BI-RADS 5 | 10 (21.7) | 4 (8.7) | 10 (21.7) | 0 | ||
| Total | 14 (30.4) | 32 (69.6) | 14 (30.4) | 32 (69.6) | 14 (30.4) | 32 (69.6) |
Abbreviations: DM, digital mammography; DBT, digital breast tomosynthesis; US,ultrasound, BI-RADS, breast imaging-reporting and data system.
a Data are presented as number and percentage.
b The BI-RADS categories of breast lesions are shown in Table 2 for each screening modality.
The diagnostic performance of all modalities was examined alone and in combination in this study. The sensitivity and specificity of DM alone were estimated at 42% and 87%, respectively. DBT detected lesions at 92% sensitivity and 68% specificity. The sensitivity and specificity of US alone were 100% and 62%, respectively (Table 3). The modality with the highest sensitivity for the detection of malignant lesions was US (100%) (95% CI: 76.84 - 100). However, there was no significant difference in the sensitivity of US and DBT in detecting malignant lesions (P = 0.999).
| Confusion matrix (TP–FP/FN–TN) | Statistical diagnostic measures (95% CI) | ||||
|---|---|---|---|---|---|
| Sensitivity | Specificity | PPV | NPV | ||
| Imaging modalities | |||||
| DM | 6 – 4 / 8 – 28 | 42.86 (17.66 – 71.14) | 87.50 (71.01–96.49) | 60 (33.34 – 81.18) | 77.78 (68.58 – 84.88) |
| DBT | 13 – 10 / 1 – 22 | 92.86 (66.13 – 9.82) | 68.75 (49.99–3.88) | 56.52 (43.25 – 8.92) | 95.65 (76.64 – 99.33) |
| US | 14 – 12 / 0 – 20 | 100 (76.84 – 100) | 62.50 (43.69–78.90) | 53.85 (42.72 – 64.60) | 100 (100 – 100) |
| P-value a | 0.016 b | 0.070 | 0.789 | 0.024 b | |
| P-value c | 0.007 b | 0.013 b | 0.646 | 0.016 b | |
| P-value d | 0.999 | 0.687 | 0.646 | 0.341 | |
| Combined imaging modalities | |||||
| DM + DBT | 13 – 10 / 1 – 22 | 92.86 (66.13 – 99.82) | 68.75 (49.99 – 83.88) | 56.52 (43.25 – 68.92) | 95.65 (76.64 – 99.33) |
| DM + US | 14 – 12 / 0 – 20 | 100 (76.84 – 100) | 62.50 (43.69 – 78.90) | 53.85 (42.72–64.60) | 100 (100 – 100) |
| DM + DBT + US | 14 – 15 / 0 – 17 | 100 (76.84 – 100) | 53.12 (34.74 – 70.91) | 48.28 (39.23 – 57.44) | 100 (100 – 100) |
| P-value e | 0.999 | 0.687 | 0.646 | 0.341 | |
| P-value f | 0.999 | 0.063 | 0.090 | 0.379 | |
| P-value g | NA | 0.250 | 0.082 | NA | |
Abbreviations: DM, digital mammography; DBT, digital breast tomosynthesis; US, ultrasound; TP, true positive; FP, false positive; FN, false negative; TN, true negative; PPV, positive predictive value; NPV, negative predictive value; NA, not available; 95% CI, 95% confidence interval.
a P-value for comparison of DM vs. DBT.
b P < 0.05 is considered statistically significant.
c P-value for comparison of DM vs. US.
d P-value for comparison of US vs. DBT.
e P-value for comparison of DM + DBT vs. DM + US.
f P-value for comparison of DM + DBT vs. DM + DBT + US.
gP-value for comparison of DM + US vs. DM + DBT + US.
Comparison of DM with DBT showed that the sensitivity of DBT was significantly higher than that of DM (P = 0.016). Also, based on the comparison of DM with US, the sensitivity of US was higher than that of DM (P = 0.07). Regarding specificity, DM showed a higher specificity than US (P = 0.013). The NPV of US was significantly higher than that of DM (P = 0.016). Besides, the NPV of DBT was significantly higher than that of DM (P = 0.024). However, there was no significant difference in terms of specificity between DM and DBT or between DBT and US (P = 0.070 and P = 0.687, respectively).
The diagnostic performance of the combination of two modalities (DM with DBT and US) and three modalities (DM, DBT, and US) is presented in Table 4. There was no significant difference in terms of diagnostic performance between the combined imaging techniques in the detection of malignant lesions (P > 0.05). In the comparison of US with DBT, US showed higher sensitivity and lower specificity than DBT (Table 4).
| ROC curve analysis | Diagnostic indices (95% CI) | |||||
|---|---|---|---|---|---|---|
| AUC (95% CI) | P-value | Sensitivity | Specificity | PPV | NPV | |
| DBT | ||||||
| Non-dense | 0.663 (0.370 – 0.885) | 0.271 | 100 (59 – 100) | 42.86 (9.9 – 81.6) | 63.6 (48 – 76.9) | 100 (100 – 100) |
| Dense | 0.920 (0.768 – 0.986) | < 0.001a | 85.71 (42.1 – 99.6) | 96 (79.6 – 99.9) | 85.7 (46.2 – 97.7) | 96 (79.6 – 99.3) |
| US | ||||||
| Non-dense | 0.918 (0.648–0.997) | < 0.001a | 71.43 (29 – 96.3) | 100 (59 – 100) | 100 (100 – 100) | 77.8 (52 – 91.9) |
| Dense | 0.954 (0.816–0.997) | < 0.001a | 71.43 (29 – 96.3) | 100 (86.3 – 100) | 100 (100 – 100) | 92.6 (79.5 – 97.6) |
Abbreviations: DBT, digital breast tomosynthesis; US, ultrasound; AUC, area under the ROC curve; PPV, positive predictive value; NPV, negative predictive value; 95% CI, 95% confidence interval.
aP < 0.05 is considered statistically significant.
Moreover, the effect of breast density on the diagnostic performance of modalities was investigated (Table 5). The specificity of DBT was significantly high in dense breasts (P < 0.001) (Table 5). However, there was no significant difference regarding the diagnostic accuracy of US between dense and non-dense breasts; the diagnostic accuracy was high for both types of breasts (P < 0.001) (Table 5). Based on the results, the sensitivity of DBT for malignant lesions (85.71%; CI: 42.1 - 99.6) was higher than US (71.43%) in the dense breast group.
| DM | Total | |||
|---|---|---|---|---|
| BI-RADS 0 | BI-RADS 3 | BI-RADS 4 | ||
| DBT | ||||
| BI-RADS 1 | 10 (21.7) U | 10 (21.7) | ||
| BI-RADS 2 | 11 (23.9) U | 1 (2.2) U | 12 (26.1) | |
| BI-RADS 3 | 1 (2.2) S | 1 (2.2) | ||
| BI-RADS 4 | 3 (6.5) D | 3 (6.5) D | 3 (6.5) S | 9 (19.6) |
| BI-RADS 5 | 4 (8.7) D | 4 (8.7) D | 6 (8.7) D | 14 (30.4) |
| US | ||||
| BI-RADS 1 | 10 (21.7) U | 10 (21.7) | ||
| BI-RADS 2 | 6 (13) U | 6 (13) | ||
| BI-RADS 3 | 3 (6.5) S | 1 (2.2) U | 4 (8.7) | |
| BI-RADS 4 | 5 (10.9) D | 8 (17.4) D | 3 (6.5) S | 16 (34.8) |
| BI-RADS 5 | 2 (4.3) D | 2 (4.3) D | 6 (13) D | 10 (21.7) |
| Total | 7 (15.2) | 29 (63) | 10 (21.7) | 46 (100) |
Abbreviations: DM, digital mammography; DBT, digital breast tomosynthesis; US, ultrasound; BI-RADS, breast imaging-reporting and data system.
a Data are presented as number and percentage.
b The superscript capital letters indicate whether DBT and US upgrade (U), downgrade (D), or show the same grade (S) as DM for the lesion.
Besides, the effect of DBT and US on the BI-RADS lesion category was evaluated in this study (Figures 1 and 2). The BI-RADS category of 22 lesions was downgraded, while 20 lesions were upgraded; also, the BI-RADS category of four lesions did not change after evaluation with DBT. By investigating the effect of US on BI-RADS categorization, it was found that the BI-RADS category of 17 lesions was downgraded, while it was upgraded in 23 lesions; it remained unchanged in six lesions (Table 5).
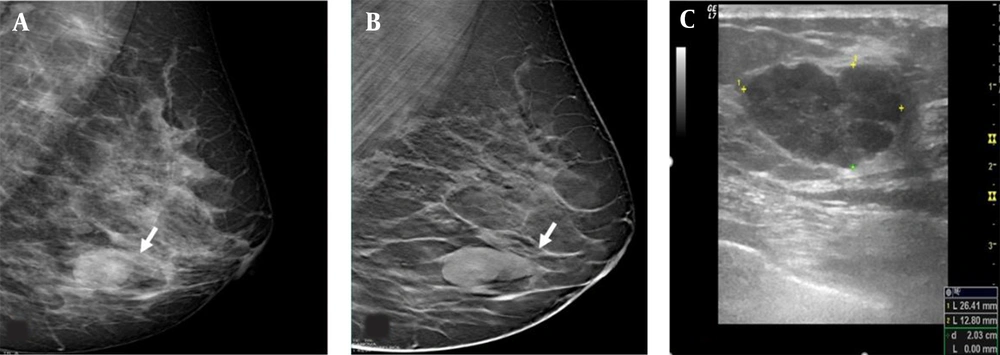
The mediolateral oblique view of digital mammography (DM) (A) reveals a nodular lesion, which is classified as breast imaging-reporting and data system (BIRADS) 0 (arrow) in the left lower breast. The mediolateral oblique view of digital breast tomosynthesis (DMT) (B) reveals well-defined contours of the lesion. On ultrasonography (C), a well circumscribed lesion is classified as category 4a, because it is palpable, and its size increases in the follow-ups. The lesion is histopathologically confirmed as usual ductal hyperplasia.
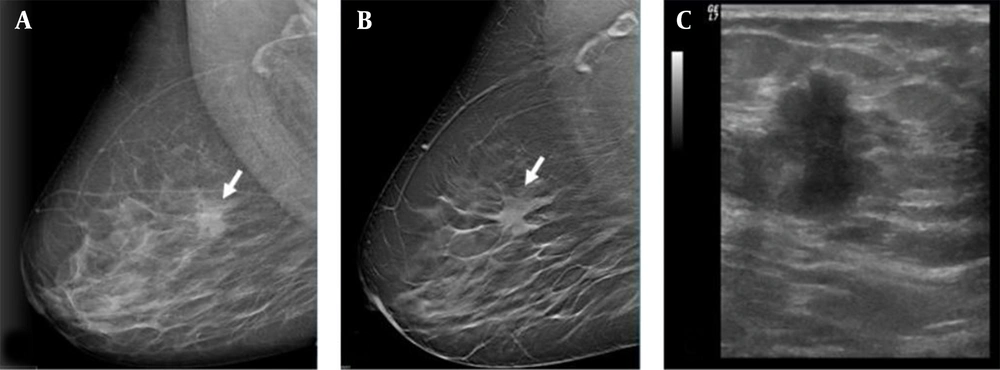
The mediolateral oblique view of digital mammography (DM). A, A focal asymmetrical density can be seen (arrow). The mediolateral oblique view of digital breast tomosynthesis (DBT) (B) shows the spiculated margin of the lesion, classified as category 5. On ultrasonography (C), a spiculated, vertically orientated lesion is classified as category 5. The mass is confirmed as invasive ductal carcinoma.
5. Discussion
DM is the most effective tool for screening and detecting breast cancer. In a recent population-based mammography screening program in Turkey, the incidence of breast cancer was estimated at 0.48% by DM screening (7). However, because dense breast tissue decreases the mammographic sensitivity and masks the findings of mass lesion, diagnosis of breast cancer can be challenging (8).
DM can detect almost all cancers in women with fatty breasts; however, its sensitivity decreases to 48% in women with dense breast tissue (9). Many researchers have accepted dense breast tissue as an independent risk factor due to poor DM visibility. Therefore, additional imaging methods are needed to increase the mammographic sensitivity. In many studies, it is known that additional imaging studies increase the detection rate of breast cancer, especially in dense breasts (8, 10, 11). On the other hand, integration of alternative modalities, such as US and DBT, in DM can help detect cancers more accurately in patients with dense breast tissue. In the present study, analysis of the contribution of DBT and US to DM showed that both US and DBT increased the diagnostic accuracy.
The US examination is a cost-effective, radiation-free, and reproducible method for the examination of breasts and indeterminate breast lesions. It is a useful modality for detecting lesions located in dense breasts when mammography cannot detect lesions (12). In this regard, Weigert and Steenbergen reported that US has an additional contribution of 0.3% to the screening of patients with dense breast parenchyma and normal DM results. Therefore, US was employed to establish a cancer diagnosis for patients with dense breast parenchyma (13). Moreover, Kim et al. (6) found that DBT had lower sensitivity than US in women with dense breasts tissue. Also, many studies have reported that combination of US with DM can help diagnose cancers more accurately in high- or average-risk women with dense breasts (6, 8).
In another study comparing DBT with US, Thibault et al. (14) reported that DBT could not outperform US. In the present study, US showed higher sensitivity for indeterminate lesions compared to DM. By incorporating US into DM for detecting lesions, the diagnostic accuracy significantly increased. Research shows that US increases the cancer detection rates, especially in dense breasts. In the present study, the integration of US in DM for both dense and non-dense breasts significantly contributed to the diagnostic accuracy and lesion characterization. Therefore, regardless of the parenchymal density, US increases the rate of breast cancer diagnosis. It should be noted that US is a subjective, operator-dependent examination, which cannot show microcalcifications and may increase the number of patients undergoing biopsy (8, 9, 14). In our study, the integration of US into DM did not upgrade the category of benign lesions; therefore, it did not lead to any unnecessary biopsy.
DBT allows for the examination of masses, parenchymal distortions, and asymmetric densities in multiple slices and angles, which can in turn reduce the false positive rates and short-term follow-ups (15). Also, downgrading is particularly important in the BI-RADS 0, 3, and 4 lesions since it reduces the recall rate. According to the ACR, BI-RADS category 0 does not establish a definitive diagnosis, and further imaging studies are needed. Also, BI-RADS category 3 indicates a probably benign tumor, which requires a follow-up schedule until stability is achieved for at least two years (16). Overall, it seems that failure to downgrade the BI-RADS category from 3 to 2 leads to an increased recall rate. Besides, BI-RADS 4 lesions are associated with multiple malignancy risks and may not demonstrate the characteristic morphology of malignant mass lesions (16).
Recent studies have mostly included BI-RADS category 0 patients and found DBT to be superior to US (15, 17). In the present study, indeterminate lesions (BI-RADS 0, 3, and 4) were investigated. The majority of our patients were classified as BI-RADS categories 0, 3, and 4. The integration of DBT into DM increased the diagnostic accuracy, and DBT showed a higher detection sensitivity than DM. Besides, the sensitivity of DBT for dense breasts was higher than US. In another study, it was reported that performing DBT after DM for indeterminate lesions detected by DM may help identify the location of the lesion more easily and decrease the recall rate (18).
In a recent study, Basha et al. (19) revealed that a combination of DM and DBT increased the accuracy of diagnosis in cases of indeterminate lesions (BI-RADS 0, 3, and 4). They also found that use of both modalities reduced the number of misdiagnosed lesions compared to DM and DBT alone (19). In another study, use of DBT also reduced the BI-RADS 3 lesions (probably benign category), and the need for unnecessary short-term follow-ups was eliminated (20). Basha et al. (19) reported that all 60 lesions, upgrading from BI-RADS 4 to 5 based on DBT, were malignant. They also reported that DBT caused a significant reduction in the number of BI-RADS 3 and BI-RADS 4 lesions (19).
In the current study, three lesions that were upgraded from BI-RADS 4 to BI-RADS 5 were considered as malignant. Bahrs et al. revealed that DBT downgraded BI-RADS 3 lesions to BI-RADS 1 or 2 lesions in almost 57% of the patients, especially in the presence of focal asymmetries (21). Since DM shows focal asymmetric densities only from a limited angle view, it may produce uncertain or false-positive results; therefore, short-term follow-up is necessary for confirming benignity.
In another recent study, the combination of DBT with DM reduced BI-RADS 3 lesions by 23.7% compared to DM alone (18). Reduced patient anxiety, decreased false positivity, and increased cost-efficiency were among the secondary advantages of downgrading probably benign findings to benign. In our study, DBT reduced the number of BI-RADS 3 lesions by downgrading or upgrading the BI-RADS category of lesions, thereby eliminating the need for unnecessary short-term follow-ups. Based on the present results, the integration of DBT and US into DM did not affect the diagnostic performance.
The present study had some limitations. First, it was performed based on the DM data, and the researchers were not entirely blinded when evaluating the DBT images. Another limitation of this study was the relatively small sample size. Consequently, we compared the diagnostic accuracy of DBT and US when both modalities were added to DM, which significantly increased the diagnostic accuracy.
In conclusion, US is a useful modality, regardless of the parenchyma pattern. The contribution of DBT was higher in patients with dense breast parenchyma compared to US. In indeterminate lesions, where DM has a low lesion detection sensitivity, the integration of easily accessible US may be prioritized. Also, if US is insufficient in dense breasts, DBT may help define the lesion.