1. Background
It is difficult to correctly diagnose transient episodes of loss of consciousness and tone if it is a seizure or cardiac syncope (1). Syncope is one of the most common complaints in children. Nearly 15% of patients under the age of 18 have experienced a period of syncope (2). Syncope is defined as the temporary loss of consciousness and postural tone in pediatrics, which could seriously affect the patient’s quality of life; it is characterized by sudden onset, short periods, and spontaneous remission (3, 4).
Syncope has a wide range of prodromal symptoms, including nausea, epigastric discomfort, tunnel and blurred vision, dizziness, palpitation, pallor, and weakness (5, 6). Different clinical presentations of syncope in children make the diagnosis difficult. In most cases, unclear causes for syncope are another challenge, putting financial pressure and concerning parents (7).
There are various reasons for syncope, and benign causes are the most important ones (8). Cardiovascular and non-cardiovascular are 2 general classifications for syncope. Cardiovascular syncope includes vasodepressor syncope, vasovagal, cardiac syncope, and vascular syncope. An essential step in managing patients is to rule out cardiac syncope with history taking, complete physical examination, electrocardiography, and other workups (9, 10). Non-cardiovascular syncope causes are hypoxia, hypoglycemia, seizure, vertigo, hyperventilation, migraine, and drugs (11).
Vasovagal syncope (VVS) is the reason for approximately 60% - 80% of neurally mediated syncopes, which is the most common cause of syncope (2). The head-up tilt test (HUTT) is a unique and helpful method to induce neurally mediated syncope. The head-up tilt test confirms the diagnosis in suspected patients and patients with inadequate data and a history of syncope. The head-up tilt test is an excellent tool to help us distinguish between epilepsy and syncope (12, 13).
Similarities between symptoms and signs of epilepsy and syncope make distinguishing between them difficult. Some syncope cases, especially convulsive syncope, are misdiagnosed as epilepsy (14, 15). Nearly 20% - 30% of patients diagnosed with epilepsy do not have epilepsy and should be classified as neuro-cardiogenic syncope. Also, 25.7% of patients diagnosed with epilepsy have a positive HUTT and were known as VVS (16).
Today, the diagnosis of syncope is based on history taking and HUTT. Moreover, the diagnosis of epilepsy is based on the clinical history and electroencephalogram (EEG). The Calgary score and modified Calgary score are other methods to distinguish epilepsy from syncope with acceptable sensitivity. Calgary has 6 questions about the anticipation of epilepsy and 3 questions about syncope prediction (17).
2. Objectives
This study aimed to differentiate between epileptic events and reflex syncope (any different type of syncope) using the head-upright tilt test.
3. Methods
Fifty-nine patients who had poor response to seizure treatment, and had triggering factors, which put epilepsy diagnosis under question, were enrolled for more evaluations. This group suspected to have syncope or convulsive syncope based on follow-up visits, taking a more detailed history or reviewing video clips of the event. The study was conducted at Mofid Children’s Hospital for 2 years.
We performed standardized essential evaluations for all patients. There were a complete history taking, information identification, physical and neurological examination, and patients referring to the pediatric cardiology clinic for more evaluations. Our colleagues measured pulse rate and blood pressure (BP) in supine and upright positions and got 12-lead electrocardiography from all patients. All basic metabolic and electrolyte workups were normal.
Each patient lay on the test table and was secured by foot straps and body belts. An intravenous line was fixed, and a prepared isotonic serum was available for possible loss of intravenous volume. The heart rate (HR) and BP were monitored throughout the test in 1-minute intervals. The BP and HR used in analyses were recorded before initiation and 5 minutes after being stable for at least 5 minutes in a 60° angle upright position. After initial preparations, the table was incrementally tilted to a 60° angle in less than 5 minutes; then, the position remained the same for 30 minutes or as long as the patient developed syncope. No provocation was used due to the patients’ age group.
Essential cardiac and basic vital signs monitoring (such as temperature, pulse rate, and respiratory rate) was performed for each patient. We continuously monitored the vital sign and electrocardiography at 1-minute intervals and recorded their information.
Whenever the patient’s symptoms progressed and were accompanied by bradycardia, hypotension, or both, we discontinued the tilt test and turned the patient back to the supine position. In this condition, we reported positive HUTT. We turned the patient back to the supine position and reported the test as negative if there was no progress in symptoms after 40 minutes.
4. Results
The tilt test was positive in 26 cases (44%) and negative in 33 cases (56%). There were 15 girls (57.69%) and 11 boys (42.31%) in the positive group, with a mean age of 11.5 years. Twenty-two girls (66.66%) and 11 boys (33.33%) were in the negative group, with a mean age of 12.3 years.
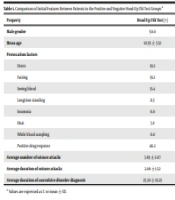
True syncope was the chief complaint in 13 cases (50%) in the positive group and 4 cases (12.1%) in the negative group. Based on their histories and examinations, the head-upright tilt test was indicated for all subjects. The mean duration of symptoms was 35 and 38 months in the positive and negative groups, respectively. The provocative factors in the positive group were stress in 19.2%, fasting in 15.3%, seeing blood in 15.3%, longtime standing in 2.6%, and insomnia in 2.6% of cases in the positive group. Provocative factors in the negative group were stress in 9.09%, fasting in 12.1%, seeing blood in 12.1%, longtime standing in 6.06%, and insomnia in 3.03% of cases (Table 1).
| Property | Head-Up Tilt Test (+) | Head-Up Tilt Test (-) | P Value |
|---|---|---|---|
| Male gender | 50.0 | 27.3 | 0.070 |
| Mean age | 10.59 ± 3.52 | 10.50 ± 3.88 | 0.922 |
| Provocation factors | |||
| Stress | 19.2 | 12.1 | 0.523 |
| Fasting | 19.2 | 9.1 | 0.112 |
| Seeing blood | 15.4 | 9.1 | 0.109 |
| Longtime standing | 11.5 | 0.0 | 0.096 |
| Insomnia | 0.0 | 3.0 | 0.978 |
| Heat | 3.8 | 3.0 | 0.834 |
| While blood sampling | 0.0 | 3.0 | 0.978 |
| Positive drug response | 46.2 | 60.6 | 0.269 |
| Average number of seizure attacks | 3.69 ± 2.07 | 3.76 ± 3.43 | 0.932 |
| Average duration of seizure attacks | 2.06 ± 1.52 | 1.98 ± 1.71 | 0.468 |
| Average duration of convulsive disorder diagnosis | 25.30 ± 19.23 | 24.75 ± 21.68 | 0.710 |
a Values are expressed as % or mean ± SD.
During the HUTT, HR increased between 0 and 50 bpm and 0 to 30 bpm in the positive and negative groups. Among 26 patients with a positive tilt test, 17 (65.3%) had VVS, and 9 (34.6%) had cardioinhibitory plus VVS (mixed type).
The average duration of the tilt table test was 10 minutes, to consider it positive. Associated symptoms were paleness in 11 cases (42.2%), dizziness in 8 cases (30.5%), weakness in 7 cases (26.9%), falling in 19 cases (72.9%), headache in 3 cases (11.9%), decreased level of consciousness in 16 cases (61.5%), and clonic movement in 4 cases (15.38%).
Ten patients from the negative group received sublingual nitroglycerin as a stimulant during the HUTT. There were no specific changes in these patients. During the test, 2 patients showed seizure-like movements such as clonic movements and a decrease in the level of consciousness. In these patients, tests stopped, and EEG was performed, which did not show any specific points regarding the seizure. Patients were followed up with the diagnosis of psychogenic seizure.
Also, in the positive group, a patient was diagnosed with VVS due to clonidine side effects (drug-related syncope). Attacks were stopped after discontinuing clonidine during a 1-year follow-up.
Most anti-seizure drugs used in patients were sodium-valproate in 71.9%, phenobarbital in 18.8%, carbamazepine in 15.6%, vigabatrin in 3.1%, risperidone in 3.1%, and clonidine in 1.7% of patients.
About 26.9% and 15.5% of patients had a positive family history of convulsive disorders in the positive and negative groups, respectively. ECG was normal in all patients.
Forty-nine patients had brain imaging, and 14 had accidental findings, such as subarachnoid cyst and Chiari malformation, increasing perivascular space, and nonspecific signal change in white matter. Electroencephalogram was abnormal in 12 cases (46.1%) in the positive group and 10 cases (30.3%) in the negative group. Epileptiform sharp waves were the most prevalent in the parieto-occipital region (27.2%), and other abnormal findings include scattered sharp waves, slow paroxysmal waves, and beta excess activity in patients on anticonvulsant or benzodiazepines (Table 2).
| Property | Head-Up Tilt Test (+) | Head-Up Tilt Test (-) | P Value |
|---|---|---|---|
| Abnormal MRI findings | 23.1 | 24.2 | 0.984 |
| Electroencephalogram findings | |||
| Normal | 61.5 | 63.6 | 0.432 |
| Focal epileptiform sharp wave | 34.6 | 9.1 | 0.009 |
| Paroxysmal sharp wave | 3.8 | 27.3 | 0.010 |
a Values are expressed as %.
5. Discussion
The result of the present study indicates the advantage of HUTT in differentiating syncope from epileptic seizures. The test result of 44% of the children in this study was positive, and they had symptoms of syncope. Two patients had clonic seizures and loss of consciousness during the test. The head-up tilt test can be helpful in children with repeated paroxysmal attacks, normal EEG, and a lack of suitable response to antiepileptic drugs (AED).
Although syncope and pre-syncope are common in children, they have low mortality and complications (18, 19). Convulsive movements and recurrent loss of consciousness can be considered epileptic events, especially in the presence of an abnormal EEG, while, in fact, it is a syncope or pre-syncope event.
In many cases, retaking the medical history of the patient’s repeated convulsions and monitoring can be helpful. However, an accurate description of attacks and access to video monitoring (with video EEG monitoring (VEM)) is needed, which is not always available due to costs. In such cases, less expensive and more functional tools, such as a tilt table test, can be helpful (20-22).
Antiepileptic drugs were tapered for these patients who were diagnosed with VVS, and they did not show any seizures after 18 ± 6 months of follow-up. When considering the time elapsed between medication withdrawal and seizure recurrence, 23.3% relapsed during weaning or within a month of medication discontinuation. Cumulative relapse rates were 53.3% of children within 6 months, 66.7% during the first year, and 90% within 3 years (23). A recent official report from the International League Against Epilepsy (ILAE) suggests that before starting treatment, it is imperative that the diagnosis of epilepsy be definite. It is also critical before prescribing antiepileptic medicines that the proposed benefits of treatment are greater than the potential consequences of treatment (24).
Stereotypical and tonic gaze is one of the reasons why syncope and pre-syncope are misdiagnosed for convulsive seizures. Also, differences between epileptic convulsions and convulsive syncope may be difficult for observers and physicians. The pathology of syncope and pre-syncope disorder is not clear (25-27). Despite similar symptoms in syncope and convulsive seizures, there are sometimes differentiating items in medical history and patients’ symptoms. These items include perspiration, nausea, vomiting, and pallor prior to the patient’s movements.
In similar studies, HUTT was positive in 40% to 70% of patients with syncope and pre-syncope compared with 44% in our study. Unlike other studies, the rate of positive response did not increase with subligual nitroglycerin TNG in our study (28, 29).
If the distinction between seizure and syncope is not clear, despite the patient’s medical history and EEG, probably unnecessary anticonvulsants will be prescribed for them and lead to strategic errors (4, 30). In our study, all patients had interictal EEG, and 37.5% were reported as abnormal. Half of the patients diagnosed with epilepsy and receiving anticonvulsants had positive HUTT results. As we know, HUTT is a complementary test, and the patients must be selected correctly to increase its predicting value. Although the tilt test is effective in differentiating cardiogenic syncope from neutrally mediated loss of consciousness, it has moderate specificity and sensitivity. On the other hand, the positive tilt test is not the reason for implanting a cardiac pacemaker; however, a detailed history and electrocardiographic studies, in addition to the tilt test, are determinants of deciding to implant a pacemaker in patients with loss of consciousness.
In cases that are resistant to seizures or are atypical, cardiologists and neurologists need to work together to accurately diagnose the type of disease. This cooperation is important in cases such as autonomic neuropathies, pseudosyncope, and pseudoseizures.
In this study, we would like to underline that despite the detailed, careful, evidence-based history taking, complete examination, and appropriate workups (as well as considering that VEM is an excellent but not available and also expensive test), distinguishing between seizures and syncope is still a big dilemma. Whereas HUTT can be useful in distinguishing syncope from seizure but cannot be useful in differentiating other types of a decrease in loss of consciousness LOC levels such as hypoglycemia, keep in mind that HUTT is an accessible test that requires less skill and time to interpret and has a cost benefit for patients, insurance companies, and health care system.
A number of patients and their parents did not cooperate properly to perform HUTT. In addition to the small sample size, our participants were highly selected by experienced pediatric neurologists, and this bias in case selection led to the high number of positive head-upright tilt test results in our study.
5.1. Conclusions
Our study showed that HUTT is a non-invasive test that can be a good modality for early and proper diagnosis in children with a history of poorly controlled epilepsy or patients with poor response to treatment and non-diagnostic EEG.