1. Background
Cancer is currently one of the deadliest diseases in the world, with numerous risk determinants such as age, diet, genetics, and environmental factors. Because it is difficult to treat cancer and due to the possibility for metastasis and relapse, there is an emerging obligation to develop new strategies for cancer therapy (1, 2). Thereafter, there is a great deal of interest in discovering new anti-cancer agents from natural sources (3).
In search for new substances with potential anti-cancer functions, natural products with many pharmacologically dynamic characteristics have been discovered and used to treat a large number of human diseases. Natural products are bioactive compounds that play an important role in inhibiting tumor growth (4). Bacteria, fungi, and plants are the main sources of the natural products and metabolites that have different biological properties, such as antitumor function (5). Although primary metabolites achieve many biochemical and physiological functions of live cells, secondary pharmaceutical metabolites under opposite circumstances are essential for survival. Bacteria are engines of ecosystems and constitute an extraordinary percentage of the organisms living in the ocean (4, 6). Marine bacteria, with an unexpectedly lower group of taxa, exhibit a broad spectrum of secondary metabolites that are considered to be the main sources of compounds with antimicrobial, antiviral, cytostatic, immunosuppressive, and insecticidal properties, as well as food supplements on earth (7-9). These organisms have contributed a significant role in the discovery and production of new biomolecules (10).
The secondary metabolites of marine bacteria are potential sources of bioactive substances that are classified as alkaloids, polyketides, polycyclic aromatic hydrocarbons, and nonribosomal peptides (NRPs). Marine bacteria are the sources of 70% of discovered polyketides and nonribosomal peptides (NRPs) and are common in the Phyla of Proteobacteria, Actinobacteria, Firmicutes, and Cyanobacteria (11, 12). Although these small natural products exhibit enormous diversity in their chemical structure, they can role as a wide range of biologically active agents such as toxins, siderophores, pigments, antibiotics, cytostatics, and immunosuppressants (10). As secondary bioactive metabolites, NRPs are synthesized by a series of enzymatic transformations, employing a diverse set of precursors on a large nonribosomal peptide synthetase (NRPS) (13).
Bacteria, cyanobacteria, and fungi are the sources of NRPS that uses both proteogenic and non-proteogenic amino acids as the building blocks of the growing peptide chain (13, 14). Polyketides have intermittent carbonyl and methylene groups and are synthesized by polyketide synthase (PKS) enzymatic complexes. Polyketide synthase enzymes are currently classified into three types that differ in the organization of their catalytic domains. The type I PKS class includes multi-domain enzymes structured in modules for the recognition, activation, and condensation of acyl-CoA in a similar way to fatty acid biosynthesis. On the other hand, PKS type II enzymes are organized in multi-enzymatic complexes, and PKS type III class mainly covers reducing enzymes (10, 12). Furthermore, due to structural and functional similarities between the enzymes responsible for the synthesis of NRP and polyketide (i.e., NRPS and PKS), hybrid peptide-polyketide products are biosynthetic products offering a wider variety of secondary metabolites (15). Studies on the biosynthetic mechanisms of polyketide and NRP by innovative genetic and biochemical techniques have revealed groups of related gene families and clusters distributed in non-uniform bacteria. Furthermore, the rapid reduction in the cost of genome sequencing now allows for the discovery of the gene clusters encoding these compounds (16, 17). Therefore, the availability of complete genomic sequences and the advantages of gene clustering in bacteria have opened the possibility of discovering clusters of metabolic genes that can produce new natural anti-cancer products by computational methods.
In this study, natural products with highly potent and desirable anti-cancer compounds that can be isolated from marine organisms are presented. Anti-SMASH webservers were recruited to analyze the genomic sequences of marine bacteria for the potential synthesis of bioactive compounds with anti-cancer effects. The biological and anti-cancer activities of these natural products as potential antitumor therapeutics have also been discussed.
2. Methods
2.1. Genome Annotation
In this study, the whole genome sequence analysis of previously isolated indigenous marine bacteria was used for various purposes. Feature Extract 1.2L Server (http://www.cbs.dtu.dk/services/FeatureExtract/) was employed to characterize the genome protein, tRNA, and rRNA from GenBank entries in the GenBank format (18). The Integrated Microbial Genomes & Microbiomes (IMG / M: https://img.jgi.doe.gov/m/) was assembled to conduct the genome annotation and sequence analysis of marine bacteria genomes in IMG (19).
2.2. Secondary Metabolites’ Gene Clusters Analysis
Mandatory marine bacteria with the genomic sequences defined in the National Center for Biotechnology Information (NCBI), the genome database (http://www.ncbi.nlm.nih.gov), were used in this study (20).
The bacterial genomic sequences of Salinispora arenicola, Prochlorococcus marinus, Pelagibacter ubique, Salinispora tropica, Alteromonas mediterranea, Crocosphaera watsonii, Synechococcus elongates, Rhodopirellula baltica, Alteromonas macleodii, Trichodesmium erythraeum, Marinobacter adhaerens, Blastopirellula marina, Oceanicola granulosus, Pseudooceanicola batsensis, Leeuwenhoekiella blandensis, Croceibacter atlanticus, Photobacterium angustum, and Cellulophaga algicola were attained from the NCBI marine bacteria genome pipeline. In silico identification of the secondary metabolites produced by similar gene clusters generating natural anti-cancer products was performed by the Anti-SMASH webserver (available at http://antismash.secondarymetabolites.org) that quickly identifies all well-known classes of secondary metabolite biosynthetic gene clusters and makes the NRPS/PKS functional annotation available (21). Furthermore, the chemical structure of NRPS/PKS components was predicted with sophisticated accuracy compared to current methods. Anti-SMASH allows to load sequence files of various types (FASTA, GBK, or EMBL files) or to automatically obtain the files associated with the GenBank/RefSeq access number from GenBank.
3. Results
3.1. Figures of Bacterial Genomes with Predicted Anticancer Gene Clusters
Genome figures for bacterial strains were predicted, and information on the GC content and the number of proteins, tRNA, and rRNA of the genomes were created by the Feature Extract. Annotation of the entire genomic sequences of marine bacteria, which potentially have gene clusters for the biosynthesis of secondary metabolites with anti-cancer capabilities, based on the results of the anti-SMASH webserver has been presented in Table 1.
| Type | Size (Mb) | GC% | Protein | rRNA | tRNA | Other RNA | Gene | Pseudogene | |
|---|---|---|---|---|---|---|---|---|---|
| Salinispora arenicola CNS-205 | Chr | 5.79 | 69.5 | 4,884 | 9 | 53 | 3 | 5,122 | 147 |
| Salinispora tropica CNB-440 | Chr | 5.18 | 69.5 | 4,486 | 9 | 52 | 3 | 4,713 | 163 |
| Crocosphaera watsonii WH 8501 | Un | 6.24 | 37.1 | 4,904 | 5 | 39 | 4 | 6,313 | 1,361 |
| Blastopirellula marina DSM 3645 | Un | 6.66 | 57.0 | 5,208 | 4 | 52 | 3 | 5,346 | 79 |
3.2. Bacteria with Gene Clusters Related to Antitumor Metabolites
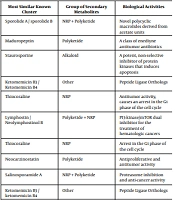
The analysis of the core genes of marine bacteria for identifying the scaffolds of secondary metabolites with anti-cancer capabilities and also their possible chemical structures, based on a subset of gene cluster types, as well as the prediction of the chemical structure and biological activity of NRPS/PKS products were performed by anti-SMASH. The analysis of marine bacterial genomes revealed gene clusters related to secondary metabolites with anti-cancer properties in four species of marine bacteria. The active derivative metabolites, their origins, targets, and mechanisms of action, and further information on these four marine bacterial species have been summarized in Table 2.
| Bacteria | Lineage | Most Similar Known Cluster | Group of Secondary Metabolites | Biological Activities |
|---|---|---|---|---|
| Salinispora arenicola CNS-205, NCBI Taxon ID: 391037 | Actinobacteria; Actinobacteria; Micromonosporales; Micromonosporaceae; Salinispora; Salinispora Arenicola; Geographical spreading: Tropical and subtropical regions | Sporolide A / sporolide B | NRP + Polyketide | Novel polycyclic macrolides derived from acetate units |
| Maduropeptin | Polyketide | A class of enediyne antitumor antibiotics | ||
| Staurosporine | Alkaloid | A potent, non-selective inhibitor of protein kinases that induces apoptosis | ||
| Ketomemicin B3 / Ketomemicin B4 | Other | Peptide Ligase Orthologs | ||
| Thiocoraline | NRP | Antitumor activity, causes an arrest in the G1 phase of the cell cycle | ||
| Lymphostin / Neolymphostinol B | Polyketide + NRP | PI3-kinase/mTOR dual inhibitor for the treatment of hematologic cancers | ||
| Thiocoraline | NRP | Arrest in the G1 phase of the cell cycle | ||
| Neocarzinostatin | Polyketide | Antiproliferative and antitumor activity | ||
| Salinispora tropica, NCBI Taxon ID: 369723 | Actinobacteria; Actinobacteria; Micromonosporales; Micromonosporaceae; Salinispora; Salinispora tropical; Geographical spreading: Tropical | Salinosporamide A | NRP + Polyketide | Proteasome inhibition and anti-cancer activity |
| Ketomemicin B3 / ketomemicin B4 | Other | Peptide Ligase Orthologs | ||
| Lomaiviticin A / C/ D /E | Polyketide: Type II + Saccharide: Hybrid/tailoring | Potent antitumor antibiotics | ||
| Lipstatin | NRP | The Beta-lactone inhibitor of human pancreatic lipase | ||
| Sporolide A / B | NRP + Polyketide: Enediyne type I | Passing phase I clinical trials as a chemotherapeutic agent | ||
| Salinilactam | Polyketide | |||
| Lymphostin / neolymphostinol B / | NRP + Polyketide | PI3-kinase/mTOR dual inhibitor for the treatment of hematologic cancers | ||
| Crocosphaera watsonii, NCBI Taxon ID: 555881 | Cyanobacteria; unclassified; Chroococcales; Aphanothecaceae; Crocosphaera; Crocosphaera watsonii; Geographical spreading: World’s open oceans | Minutissamide A /C/D | NRP | Antiproliferative activity |
| Aranazole A/ B/ C/ D | NRP + Polyketide | Halogenating Enzymes for Active Agent Synthesis, biocatalysis | ||
| Blastopirellula marina, NCBI Taxon ID: 314230 | Planctomycetes; Planctomycetia; Pirellulales; Pirellulaceae; Blastopirellula; Blastopirellula marina; Geographical spreading: Resides in eutrophic, salty, aquatic environments | nocuolin A | NRP + Polyketide | Apoptotic inducers |
These marine bacterial species were Salinispora arenicola, Salinispora tropica, Crocosphaera watsonii, and Blastopirellula marina. The genomes of two of these species (S. tropica and S. arenicola) coded eight and seven gene clusters related to known anti-cancer products of polyketides and NRP, respectively. Crocosphaera watsonii showed two gene clusters, while Blastopirellula marina showed one gene cluster encoding products with natural antitumor properties.
4. Discussion
The advent of next-generation sequencing technologies (NGS) permits the discovery of numerous clusters of genes that encode natural and biologically active products. For this reason, exploration of favorable targets within genomes by in silico genomic mining is necessary (21). Marine bacteria are major sources of numerous bioactive metabolites as anti-cancer agents. Among known organic compounds, polyketides and NRPs are undoubtedly valuable for their anti-cancer properties (15).
The results of this study demonstrated that marine bacterial species of S. arenicola, S. tropica, C. watsonii, and B. marina harbored efficient antitumor gene clusters encoding secondary metabolites with potential anti-cancer properties.
Salinispora arenicola species is an obligatory marine actinomycete found in a large geographic area in the tropical and subtropical region and contains abundant content of polyketides and NRP, which are secondary metabolites with anti-cancer features (22). Salinosporamide A (Marizomib), an NRP-polyketide hybrid marine microbial metabolite, is a potent inhibitor of 20S proteasome and induces apoptosis, and its chlorine atom is crucial for strong proteasome inhibition and antitumor activity (22, 23). Accordingly, predicted natural anti-cancer products of S. arenicola include Maduropeptin Hromophore, an antibiotic with antitumor activity, Staurosporin that mainly inhibits protein kinases through the prevention of their binding to ATP, and also ketomemycins with catalytic amino acid binding pseudodipeptide for ATP-ligase activity (23-26). Thiocoralin, a depsipeptide, is another example of bioactive products of S. arenicola, which inhibits the cell cycle at the G1 phase and reduces the progression of the S phase towards the G2/M phase (27). Neolymphostin A, a dual inhibitor of PI3-kinase/mTOR (28, 29), and chromocore Neocarzinostatin, which induces DNA double-strand breaks, are expected to inhibit DNA replication (30).
Salinispora tropica, like S. arenicola, is isolated from tropical marine sediments and belongs to the order of Actinomycetales that produces branched hyphae. There are similarities in the gene clusters linked to the Salinispora species, indicating the existence of shared metabolites in these two bacterial species. These organisms may be new sources of bioactive chemicals to be used in the treatment of various diseases such as cancers. Analysis of this species identified numerous gene clusters encoding polyketides, NRPs, and nonribosomal hybrid peptides with cancer-inhibiting capabilities. This species produces the bioactive compounds of salinilactam and lipstatin that may be useful in the treatment of cancer. Small-ringed macrolactam-like salinilactam has been shown to have a wide range of activities, e.g., bactericidal, antiviral, cytotoxic, antifungal, and antiprotozoal. Lipstatin as a potent and selective inhibitor of human pancreatic lipase, lomaiviticin A and B as potent anti-cancer antibiotics, and salinosporamide A, which is on phase I clinical trials for cancer treatment, are potential anti-cancer products of S. tropica (23, 31-35).
Crocosphaera watsonii that belongs to single-cell marine diazotrophic cyanobacteria and has been isolated from oceanic oligotrophic waters in the tropical area, is regarded as a resource for carbon and nitrogen in the ocean. Minutissamide, as a cyclic lipodecapeptide, is produced by a known gene cluster of this species and has a hypophilic amino acid residue (octadecanoic or tetradecanoic acid) that exhibits antiproliferative activity against cancer cells (36, 37).
Blastopirellula marina, a halotolerant bacterium and a representative of Planctomycetales with budding branches, is quite abundant in terrestrial and marine habitats. This bacterium influences global biogeochemistry and climate by catalyzing essential transformations in global carbon and nitrogen cycles to have an emotional exchange between the geosphere and the atmosphere. A similar gene cluster encoding oxadiazine Nocuolin A (NocA), a natural product with potential antitumor activity, in the genome of B. marina has shown cytotoxic effects against several colon cancer cell lines by inducing caspase-dependent apoptosis (38, 39).
At this time, it has been claimed that many consequential marine products are capable of being expanded into pharmaceuticals. Consequently, it is necessary to study the anti-cancer function of marine natural products in order to develop effective anti-cancer drugs for various types of cancers.
4.1. Concluding Remarks
The availability of the complete genomic sequences of microbial organisms provides the opportunity to use in silico genome analysis for secondary metabolite biosynthesis. The natural products derived from marine bacteria are sources of bioactive secondary metabolites that can emerge as a new generation of promising anti-cancer drugs. However, the therapeutic and clinical application of a promising marine product depends on the results of experimental studies. The marine ecosystem is regarded as a great source of many natural products, and the improvement of extraction procedures has boosted the quality of natural products. Among marine bacteria with known genomic sequences, the two species of S. tropica and S. arenicola are richer in the genes encoding polyketides and NRPs, which are potential sources of new natural anti-cancer products.