1. Background
Tricothecene is classified as a mycotoxin, which is produce by various species of fungi including Fusarium, Trichoderma, Myrothecium, Verticimonisporium, and Stachynotris (1). T-2 toxin, which belongs to the tricothecene group, is mainly produce by Fusarium sporotrichoedes (2), and has the highest toxicity among the toxins produced by Fusarium. In the case of oral administration in rats, T-2 toxin is mainly absorbed in the intestine. T-2 toxin causes necrotic lesions and bleeding in the stomach, intestine, liver, and kidney (3). T-2 toxin absorbs through the skin and causes local inflammation that is potentially lethal. Macroscopic evaluation indicates that in humans, severe dermal irritation caused by small amounts of T-2 toxin can potentially last for two weeks. A variety of mechanisms of action have been proposed for T-2 toxin, which reacts with the thiol groups of sulfhydryl enzymes and inhibits protein and DNA synthesis (4), as well as impairs the production of antibodies, alters membrane functions, reduces lymphocyte proliferation, and alters the maturation process of dendritic cells (5-7). Currently, the demand for herbal therapy is increasing and the amount of research being conducted on traditional herbal drugs indicates that many people around the world trust such remedies to alleviate various skin diseases (8). The genus Crataegus belongs to the family Rosaceous comprising a complex group of trees and shrubs that are native to Northern temperate zones, mostly between latitudes 30° and 50° N. Hawthorn refers to the plant Crataegus and is widely distributed throughout the Northern temperate regions of the world having approximately 280 species. Crataegus pontica is a small tree or large spiny shrub, mostly growing 6 - 10 m tall, with a dense crown. The bark is dull brown with vertical orange cracks. Leaves grow in clusters on spur shoots and most species have lobed or serrated margins. They are somewhat variable in shape, with the lobes spreading at a wide angle. Blossoms are white; petals are oval or approximately round. The fruit is berry-like in appearance, having a pome-like structure, with a golden yellow skin color. Crataegus pontica C. Koch (Persian name: zalzalak) is a member of this genus and is widely distributed throughout the western, northwestern, and central areas of Iran (9). Almost all parts of these plants are reported to possess various pharmacological properties including anti-inflammatory, gastro-protective, antimicrobial, cardio tonic, hypotensive, and hepatoprotective effects. In Iranian traditional medicine, the leaves and flowers of some Crataegus species have been used to produce decoctions for various purposes such as tonics for cardiovascular diseases, sedatives, and antianxiety agents. Hawthorn fruit has been reported to possess cardio tonic, coronary vasodilator, and hypotensive properties. Traditionally, it has been used for cardiac failure, myocardial weakness, paroxysmal tachycardia, hypertension, arteriosclerosis, and Buerger's disease. Hawthorn leaves, flowers, and berries contain a variety of bioflavonoid-like complexes that appear to be primarily responsible for the plant’s actions on the cardiac system (9, 10).
2. Objectives
The experiments described in the present study were performed to examine the healing effects of creams prepared using Crataegus pontica leaf extract on dermal toxicity induced by T-2 toxin.
3. Methods
3.1. Animals
Male and female Iranian rabbits (1.8 - 2.2 kg) were obtained from the animal house at Ahvaz Jundishapur University of Medical Sciences. Animals were housed individually in cages and allowed to feed on a standard commercial pellet diet supplemented with fresh vegetables, carrots, and tap water ad libitum. The animals were maintained under controlled conditions at 20 ± 2 °C with a 12 hours light: 12 hours dark cycle. The investigation was performed according to the Local Animal Ethics Committee guidelines for the use of experimental animals.
3.2. Extract Preparation
Fresh C. pontica leaves were collected from the Khuzestan region of Iran in September 2013. The leaves were identified at the herbarium in the department of pharmacognosy, school of pharmacy, Ahvaz, Iran. The leaves were dried in shade at room temperature, crushed into small pieces, and soaked in a 70% aqueous-ethanol solution in a large container for 3 days with occasional shaking (11). The extract was filtered through a clean cotton cloth, dried using a rotary evaporator at 40°C, and then freeze-dried. The extract yield (dry powder) was 16% w/w. The powdered extract and eucerin were then mixed at proportions of 5 to 95 m/m, 10 to 90 m/m and 15 to 85 m/m, to respectively produce C. pontica creams at concentrations of 5%, 10%, and 15%.
3.3. Experimental Schedule
T-2 toxin was supplied in a 5 mg vial, which was solubilized using ethanol (as a solvent) to a final volume of 0.6 ml before use. The hair on the animals’ lower backs around the backbone was shaved so that the areas were fully clear. A 1.5 × 1.5 cm square was drawn on the desired area using a marker. The toxin solution was topically administered to the marked region using a Hamilton micro syringe. To induce dermal toxicity, a solution of 100 µg/12 µl T-2 toxin was applied to the marked skin surface area for 2 successive days. The experiment was performed on six groups, each containing 5 rabbits. The groups were treated as follows: group 1 (positive control) was treated with toxin only; group 2 (negative control) was treated with eucerin only; group 3 was treated with 5% C. pontica cream in a eucerin base; group 4 was treated with 10% C. pontica cream; group 5 was treated with 15% C. pontica cream; and group 6 was treated with ethanol only (to evaluate the safety of the T-2 toxin solvent). All treatments commenced 48 hours after toxin administration; treatment was applied topically to the surface of the damaged areas twice per day for 7 consecutive days. During treatment of the various groups, damages due to toxicity, including blistering, inflammation, and erythematic (redness) reactions, were evaluated by means of a score ranging from +1 to +4, according to severity.
3.4. Statistical Analysis
Statistical evaluation was performed, using the Kruskal–Wallis test. P-values less than 0.05 were considered significant.
4. Results
4.1. Macroscopic Inspection
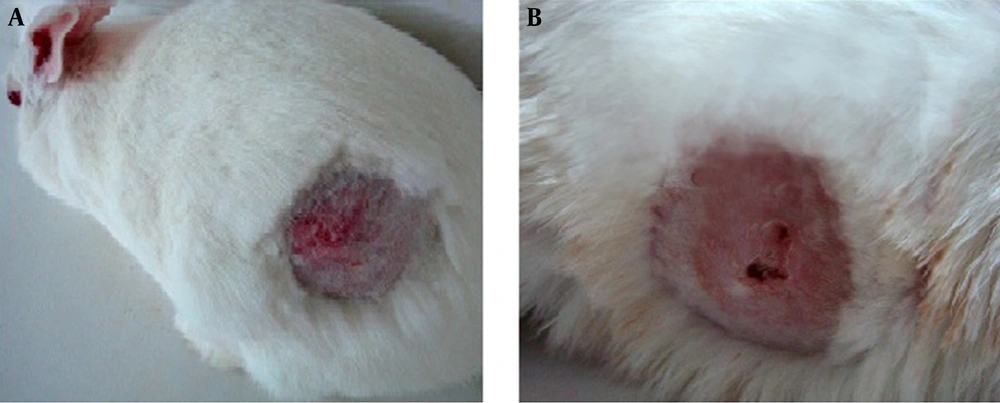
The skins of the animals in the different groups were inspected daily and the degree of healing of the lesions was recorded. In the group that was treated with ethanol, no abnormalities were observed on the initial day; therefore histological studies were not performed in this group. No animal mortality occurred during the study. In the first 48 hours after toxin application, edema, stiffness, and erythema were observed in the epidermis; furthermore, seemingly deep blisters formed in some areas (Figure 1A). On the 9th day after T-2 toxin administration, central necrosis surrounded by an erythematic reaction was observed (Figure 1B).
The macroscopic healing pattern of the lesions and skin damages began with scab formation over the wound on the 8th to 12th day after initiation of the treatment. Thereafter, the scab was removed from the lateral part of the wound or simultaneously completely removed at the end of the healing stage (Figure 2).
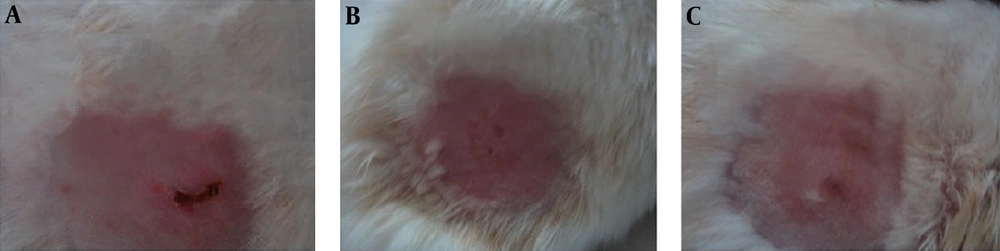
(A-C), on the 10th day of treatment. Erythema, inflammation, and partial necrosis are seen in the 5% treatment group. Erythema and inflammation are not seen in the 10% treatment group. In the 15% treatment group, there is no erythema or inflammation and normal skin and hair regrowth are observed.
4.2. Inspecting the Inflammation, Erythema, and Blisters
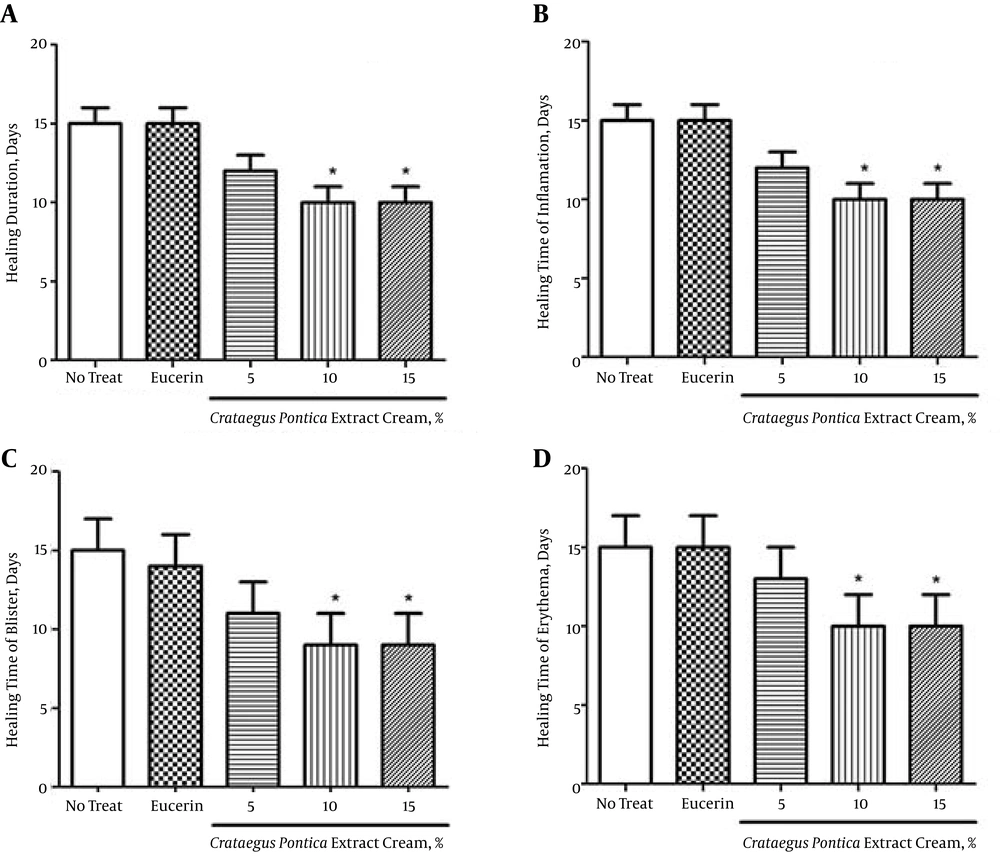
On the 3rd day of treatment, inflammation was observed in all groups that had received the toxin. There were no significant differences in inflammation, erythema, or blister healing between the treatment and non-treatment groups. Compared to the other groups, on the 10th day of the study, there was less inflammation, erythema, and blister damage in the groups treated with the 10% and 15% C. pontica creams (Figure 2).
There were no significant differences in inflammation, erythema, and blister healing between the groups treated with the 10% and 15% C. pontica creams, but a significant difference was observed compared to the eucerin group (P < 0.05) (Figure 3).
4.3. Histopathological Findings
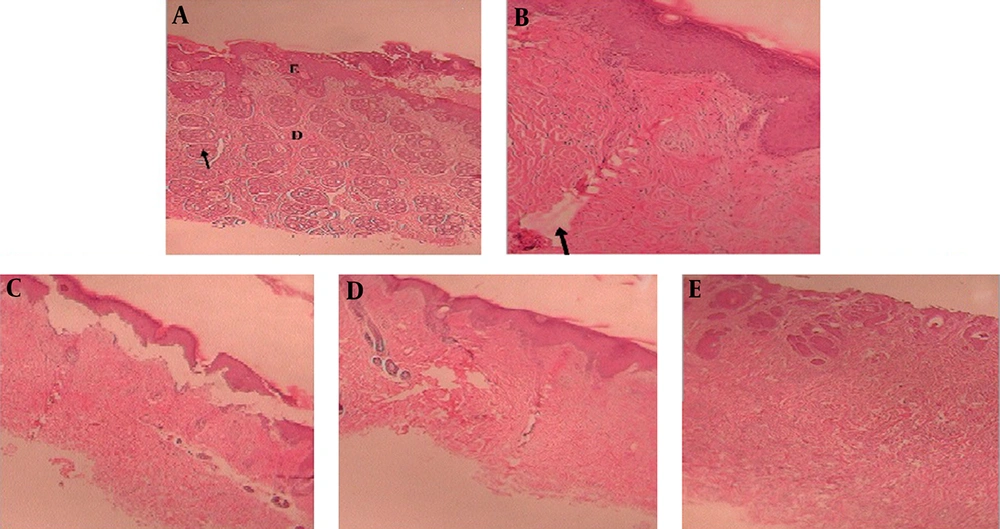
Almost 48 hours after the primary application of the toxin, primary destruction (necrosis and wound lesions) of the epidermis and vessel dilation with lymphocyte aggregation was observed in the group without epidermal treatment, indicating the initiation of the inflammation reactions (Figure 4B). Seven days after primary application of the toxin, parts of the wound lesions, in addition to dispersed granulation aggregation and inflammatory reactions, remained.
On the 10th day of treatment in the group treated with the 5% cream, no wound lesions had concisely formed in the epidermis. Furthermore, a slight layer of epidermis and dermal granulation tissue had formed, and collagen precipitation and dispersed lymphocyte aggregation was observed (Figure 4C). In the group treated with the 10% cream on the 10th day of treatment, we observed reformation of the epidermis, complete dermal granulation, tissue formation, collagen precipitation, and no inflammation (Figure 4D). In the group treated with the 15% cream, the epidermis reached the normal level and complete granulation tissue and intact fibroblast strands were observed on the 10th day of treatment (Figure 4E).
5. Discussion
A wound is a physical injury that arises from an opening or break in the skin. Wound healing is a collaborative process involving a variety of cells and matrix components interacting continually toward a common goal (12, 13). This process is characterized by three stages: inflammation, proliferation, and remodeling. The proliferative phase typically demonstrates angiogenesis, collagen deposition, granulation tissue formation, epithelialization, and wound contraction (14, 15). Trichothecenes are secondary metabolites produced by some species of fungi. More than one hundred trichothecenes have been identified in the laboratory, but only about a dozen of these compounds have been found under natural conditions (16, 17). Tricothecenes are classified according to their chemical structures, based on a ring system denominated 12, 13-epoxytrichothec-9-ene with several possible functional groups attached. Deoxynivalenol (DON) and nivalenol (NIV) are the most frequently studied trichothecenes, and diacetoxyscirpenol (DAS) and T-2 toxin are the most toxic among the non-cyclic compounds of the family (18, 19).
In the present study, in comparison to the control and eucerin-treated groups, topical application of C. pontica cream caused acceleration in wound healing as indicated by the decreased time to complete epithelization. However, the best results were obtained with the 10% and 15% C. pontica extract creams.
To further confirm the quality and maturity of the healing tissue on different days and in the different groups, we microscopically examined histopathological wound sections stained by hematoxylin and eosin (H&E). In many previous studies evaluating wound healing, parameters such as edema, fibrin clots, infiltration of white blood cells and inflammatory cells, intensity of fibroblasts, angiogenesis, texture of granulation, re-epithelization, and collagen intensity have been examined histopathologically (20, 21). In the present study, the histological findings corresponded with the observed reductions in the damaged areas. The H&E stained sections revealed that in the group treated with the 5% cream, on the 12th day of treatment, there were no wound lesions in the epidermis, epidermis and dermal granulation tissue had formed, and collagen precipitation and dispersed lymphocyte aggregation was observed. In the group treated with the 10% cream on the 10th day of treatment, there was no inflammation, and reformation of the epidermis, complete dermal granulation tissue, and collagen precipitation were observed. In the group treated with the 15% cream, normal levels of epidermis, complete granulation tissue, and intact fibroblast strands were observed on the 10th day of treatment.
There is increasing interest in the use of herbal medicines for wound healing. Flavonoids have significant impacts on wound healing and dermal protection. These components prevent blood and lymph accumulation by regulating micro vessels around the wound lesion area and thus reduce local swelling. They also improve blood circulation and enhance continuous perfusion to facilitate the healing of wound lesions (22, 23). Since the most important class of active ingredients present in Crataegus sp. is flavonoids (including hyperoside, vitexin, isoquercetin, luteolin 7-glucoside, and quercetin), it is suggested that the flavonoids present in the available extract must play a crucial role in the treatment of damage caused by T-2 toxin.
A similar study investigated the healing effects of quince seed mucilage on dermal toxicity induced by T-2 toxin. In this study, to induce dermal toxicity, 100 µg of T-2 toxin was applied to the shaved skin of rabbits. The obtained results indicate that quince seed mucilage has remarkable healing effects on dermal toxicity caused by T-2 toxin (1).
In the present study, it was observed that doses of both 10% and 15% C. pontica cream had similar effects on wound healing, therefore 10% C. pontica cream can be suggested as a potential treatment for T-2 toxin-induced dermal toxicity.
Finally, this study confirmed that C. pontica cream accelerated wound healing and the time taken for complete healing of damaged skin when compared to the negative control and eucerin only groups. The beneficial effects of the C. pontica preparation in the healing of superficial skin wounds may be attributed to its flavonoid content. Furthermore, the present study indicates that this preparation could be applied for the treatment of skin damage and may be an alternative to the use of other routine treatments.