1. Background
Meniere’s disease is an inner ear disorder caused by the accumulation of endolymphatic fluid in the cochlea and vestibular organ (1). Tinnitus, vertigo, and hearing loss are among the most common symptoms of this condition (2). Older, white, female patients are the most vulnerable group affected by Meniere’s disease, and accumulating evidence indicates that the prevalence of this disorder increases with age (3, 4). Migraine, polygenic diseases, and autoimmune disorders are suggested as the main comorbidities associated with this condition (5). Theories proposing multifactorial causes of Meniere’s disease are the most widely recognized (6).
Due to the significant impact of Meniere’s disease on the social functioning of affected individuals, various management strategies have been proposed (7). Treatment options include intratympanic gentamycin injections, intratympanic steroid injections, betahistine, and thiazide diuretics (8-11). Additionally, a sodium-restricted diet may reduce Meniere’s attacks (7). However, substantial disagreement exists within the medical community regarding the application of these treatment options.
Given the inflammatory and autoimmune nature of Meniere’s disease (12), anti-inflammatory and immunomodulatory agents such as infliximab, cepharanthine, celastrol, resveratrol, and vitamin D could potentially alleviate this syndrome (12-14). Vitamin D, a fat-soluble vitamin, exerts several biological effects, including anti-cancer, anti-aging, immunomodulatory, and anti-inflammatory activities (15). Studies suggest that vitamin D may alleviate Meniere’s disease by inhibiting the adaptive immune system and suppressing inflammation (16, 17).
A meta-analysis by Salamah et al. (18) concluded that vitamin D deficiency is associated with the etiopathology of ear diseases, including Meniere’s disease, in both adults and children. The authors noted that vitamin D deficiency is common in otolaryngology patients, and supplementation showed promising results (18). A recent case-control study by Bakhshaee et al. (17) revealed that serum concentrations of vitamin D in the Meniere’s disease group were significantly lower than those in the control group. However, the authors emphasized the need for further clinical investigation to establish the beneficial role of vitamin D in managing Meniere’s disease (17).
2. Objectives
Due to the lack of sufficient randomized clinical trials in this area, the present research project evaluated the effectiveness of vitamin D supplementation as an adjuvant therapy for vertigo and tinnitus in patients with Meniere’s disease.
3. Methods
3.1. Study Design and Participants
This double-blind, placebo-controlled, randomized clinical study was conducted on patients with Meniere’s disease to evaluate the effects of vitamin D on vertigo and tinnitus. The study design was approved by the Ethics Committee of Research, Tehran, Iran (IR.TUMS.TIPS.REC.1400.137) and was registered at the Iranian Registry of Clinical Trials (IRCT20210716051908N1). At the beginning of the study, participants read and signed an informed consent form.
Seventy patients with Meniere’s disease, aged over 18 years, with serum vitamin D levels ranging from 10 to 30 ng/mL (indicating vitamin D deficiency), were recruited. After screening, 66 eligible participants were randomized into either the intervention or placebo group. The diagnosis of Meniere’s disease was performed by an expert according to the American Academy of Otolaryngology-Head and Neck Surgery (AAO-HNS) guidelines. Patients with two or more episodes of spontaneous vertigo, each lasting at least 20 minutes to 12 hours, an audiometric test indicating sensorineural hearing loss, and clinical symptoms such as tinnitus and ear fullness were diagnosed with definite Meniere’s disease.
Exclusion criteria included pregnancy, lactation, use of medications affecting vitamin D metabolism, and the presence of liver, kidney, thyroid, parathyroid, neurological, and autoimmune diseases. Subjects with metabolic disorders affecting serum levels of vitamin D (e.g., inflammatory bowel syndrome and celiac disease) and patients with middle ear infections were also excluded. All treatments, except for betahistine (24 mg/day), were discontinued before the trial began.
3.2. Sample Size Estimation
Considering a type I error of 5% (α = 0.05) and a type II error of 0.2, and based on a previous study by Yardley and Kirby (19), the sample size was calculated to be 30 for each group. To account for potential dropouts, we added 10% to the sample size.
3.3. Randomization and Intervention
Patients were randomly assigned to either the vitamin D or placebo groups using the permuted block randomization method with four blocks. Patients in the intervention group (n = 30), in addition to receiving the standard treatment for Meniere’s disease (betahistine 24 mg/day), were supplemented weekly with vitamin D (50,000 IU, manufactured by Zahrawi Company). Given the patients’ vitamin D deficiency, we prescribed 50,000 IU of vitamin D weekly after lunch for eight weeks, following the guidelines of the Ministry of Health of Iran for the Iranian population with vitamin D deficiency (20).
Patients in the control group (n = 30), in addition to receiving the standard treatment for Meniere’s disease (betahistine 24 mg/day), were supplemented weekly with a placebo (soft gel capsules filled with water), which was identical in shape and color to the vitamin D capsules. A person with no knowledge of the study’s method and purpose coded the packages of vitamin D supplements and placebos using a list of random numbers. Thus, researchers were blinded to the types of supplements participants consumed. Study participants were also unaware of the group to which they were assigned. Adherence to the supplement regimen (taking ≥ 80% of the supplements) was assessed based on the unused capsule counts for each patient.
3.4. Study Outcomes
Changes in the number of vertigo attacks, as well as the duration and severity of vertigo, were regarded as the primary outcomes of this research. Additionally, changes in the duration and severity of dizziness were considered primary outcomes, while any alterations in tinnitus indices were secondary outcomes.
3.5. Assessment of Study Outcomes
General and medical information was collected via questionnaire at the beginning and end of the trial. Five milliliters of venous blood were drawn from each patient at the onset and conclusion of the investigation to measure serum concentrations of vitamin D using enzyme-linked immunosorbent assay (ELISA) kits. Each patient completed the Dizziness Handicap Inventory (DHI), Visual Analogue Scale (VAS), and Tinnitus Handicap Inventory (THI) questionnaires at the beginning and end of the study. These questionnaires assessed the intensity, frequency, and duration of vertigo attacks, as well as the intensity of tinnitus and their impact on the patient’s quality of life.
The VAS was used to assess the severity of vertigo, with scores ranging from zero to ten. In this questionnaire, frequency was defined as the number of vertigo attacks per month, and duration was defined as the average duration of each vertigo attack. Monthly vertigo for each patient was calculated as the duration of vertigo multiplied by the frequency of vertigo (21).
The DHI consists of 25 questions across three domains: Physical, emotional, and functional. Each question was answered with "no" (zero points), "sometimes" (2 points), or "yes" (4 points) (22). Total scores ranged from 0 (no disability) to 100 (severe disability). The THI was used to assess tinnitus, comprising 25 questions, each answered with "no" (zero points), "sometimes" (2 points), or "yes" (4 points). Higher scores indicated more severe tinnitus and greater disruption of daily activities (23).
3.6. Statistical Analysis
All statistical analyses were conducted according to the per-protocol (PP) method, considering only participants who completed the 8-week study period. The Shapiro-Wilk test was used to check the normal distribution of variables. Between- and within-group differences were evaluated using independent samples t-tests and paired samples t-tests for continuous variables with a normal distribution, respectively. Inter-group differences in qualitative variables were evaluated using the chi-square test. At the study’s conclusion, inter-group changes were tested using the two-sample t-test with equal variance. A significance level was set at a P-value of less than 0.05. Stata Statistics software (StataCorp. 2019. Stata Statistical Software: Release 16. College Station, TX: StataCorp LLC) was used for data entry and statistical analysis.
A linear mixed model was applied for the repeated measurement data from follow-up visits. Repeated-measure, mixed-effect linear regression models (22) were used to evaluate changes over time in vertigo severity, vertigo duration, vertigo attacks, DHI total, DHI emotional, DHI functional, DHI physical, THI total, THI emotional, THI functional, THI catastrophic, and serum 25 (OH) vitamin D. To assess the intervention effect as a change over time in each variable among all patients (model 1) and determine time by group interaction in each variable (model 2), evaluations were conducted. The models considered time variables and baseline fixed effects for participant characteristics.
4. Results
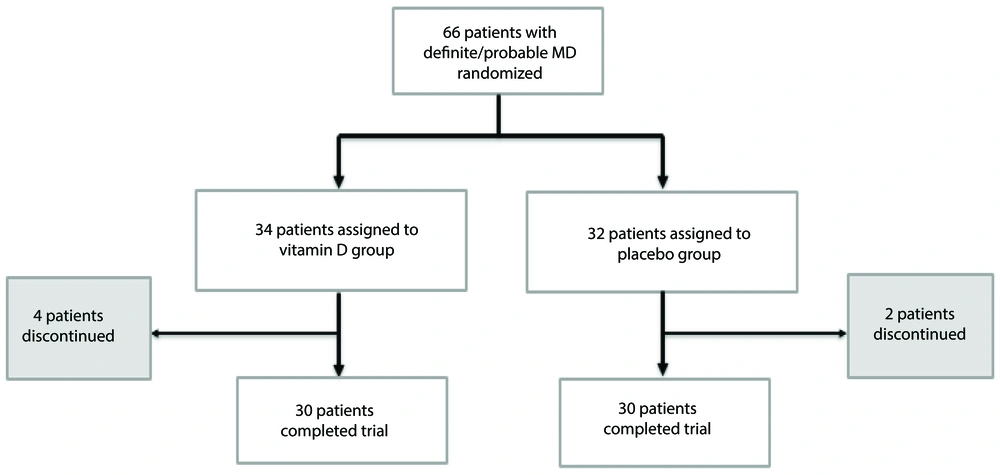
Figure 1 illustrates the flowchart of the current clinical trial. Of the 66 patients randomized, 60 subjects (30 patients in each group) completed the trial. Two patients in the placebo group and four patients in the intervention group were lost to follow-up for reasons unrelated to the interventions. There were no complaints or complications following drug/placebo use, and no toxic levels of vitamin D were detected at the end of the intervention.
Table 1 presents the baseline characteristics of participants in the vitamin D and placebo groups. The groups were well-matched in terms of age, sex distribution, smoking status, and medication use, indicating a balanced randomization process. Notably, the post-intervention vitamin D level was significantly higher in the supplementation group compared to the placebo group (P < 0.001), confirming adherence to the intervention. There was no significant difference in baseline characteristics, including age, sex, serum levels of vitamin D, smoking status, and history of betahistine, triamterene/hydrochlorothiazide, corticosteroid, and cinnarizine consumption.
| Variables | Vitamin D (n = 30) | Placebo (n = 30) | P-Value |
|---|---|---|---|
| Age (y), mean (range) | 48.00 (42.10 - 53.89) | 48.97 (44.35 - 53.59) | 0.79 |
| Vitamin D level (before) | 21.86 | 21.99 | 0.92 |
| Vitamin D level (after) | 36.94 | 20.48 | < 0.001 b |
| Sex | 0.432 | ||
| Male | 14 (46.66) | 11 (36.66) | |
| Female | 16 (53.33) | 19 (63.33) | |
| Smoking | 1.00 | ||
| Yes | 3 (10) | 3 (10) | |
| No | 27 (90) | 27 (90) | |
| Betahistine | 0.228 | ||
| Yes | 25 (83.33) | 28 (93.33) | |
| No | 5 (16.66) | 2 (6.66) | |
| Triamterene/hydrochlorothiazide | 0.152 | ||
| Yes | 6 (20) | 11 (36.66) | |
| No | 24 (80) | 19 (63.33) | |
| Corticosteroid | 0.085 | ||
| Yes | 5 (16.66) | 1 (3.33) | |
| No | 25 (83.33) | 29 (96.66) | |
| Cinnarizine | 0.542 | ||
| Yes | 8 (26.66) | 6 (20) | |
| No | 22 (73.33) | 24 (80) |
Baseline Characteristics of Vitamin D and Placebo Group a
As indicated in Table 2, within-group comparisons showed a significant decrease in the mean number of vertigo attacks, the duration and severity of vertigo, various assessed indices of dizziness, and the score of emotional THI in both study groups at the final assessment. This significant within-group comparison might suggest a placebo effect. Regarding between-group comparisons, there were no significant alterations in the mean number of vertigo attacks (P = 0.85), duration (P = 0.52), or severity (P = 0.33) of vertigo at the end of the trial after vitamin D supplementation. However, in the vitamin D group, the duration of vertigo was approximately 47 minutes less than in the placebo group. Additionally, vitamin D supplementation had no discernible effects on the results of various dizziness and tinnitus indices at the study’s conclusion compared to the control group (P > 0.05). As expected, serum levels of vitamin D were significantly higher in the intervention group compared to the placebo group at the trial’s endpoint (P < 0.001).
| Models and Variables | Vitamin D (n = 3) | Placebo (n = 30) | Δ c | P-Value | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Before | After | Δ c | P-Value | Before | After | Δ c | P-Value | |||
| Model 1: Time | ||||||||||
| Vitamin D level (ng/mL) | 21.92 ± 1.10 | - | - | - | 28.71 ± 1.10 | - | - | - | 6.78 | < 0.001 d |
| Vertigo attack | 4.16 ± 0.53 | - | - | - | 1.60 ± 0.53 | - | - | - | -2.56 | < 0.001 d |
| Vertigo duration (min) | 223.83 ± 35.88 | - | - | - | 34.18 ± 35.88 | - | - | - | -197.88 | < 0.001 d |
| Vertigo severity | 9.23 ± 0.28 | - | - | - | 4.11 ± 0.28 | - | - | - | -5.11 | < 0.001 d |
| DHI. total | 60.03 ± 2.10 | - | - | - | 25.30 ± 2.10 | - | - | - | -34.73 | < 0.001 d |
| DHI. emotional | 50.41 ± 2.16 | - | - | - | 21.08 ± 2.16 | - | - | - | -29.33 | < 0.001 d |
| DHI. funtional | 70.43 ± 2.73 | - | - | - | 30.02 ± 2.73 | - | - | - | -40.41 | < 0.001 d |
| DHI. physical | 61.90 ± 2.26 | - | - | - | 25.94 ± 2.26 | - | - | - | -35.95 | < 0.001 d |
| THI. total | 50.66 ± 3.55 | - | - | - | 44.40 ± 3.55 | - | - | - | -6.26 | 0.215 |
| THI. emotional | 49.18 ± 3.48 | - | - | - | 39.49 ± 3.48 | - | - | - | -9.69 | 0.051 |
| THI. functional | 52.57 ± 3.74 | - | - | - | 47.11 ± 3.74 | - | - | - | -5.45 | 0.305 |
| THI. catastrophic | 48.5 ± 4.40 | - | - | - | 45.75 ± 4.40 | - | - | - | -2.75 | 0.659 |
| Model 2: Time group | ||||||||||
| Vitamin D level (ng/mL) | 21.8 ± 1.12 | 36.88 ± 2.06 | 15.08 | < 0.001 e | 22.06 ± 1.08 | 20.54 ± 1.19 | -1.52 | 0.07 e | 16.6 | < 0.001 d,f |
| Vertigo attack | 4.30 ± 0.96 | 1.63 ± 0.36 | -2.67 | < 0.001 e | 4.03 ± 0.97 | 1.56 ± 0.38 | -2.47 | < 0.001 e | -0.20 | 0.85 f |
| Vertigo duration (min) | 262.33 ± 78.19 | 49.20 ± 24.73 | -213.13 | < 0.001 e | 185.33 ± 65.42 | 19.16 ± 4.58 | -166.17 | < 0.001 e | -46.96 | 0.52 f |
| Vertigo severity | 9.20 ± 0.18 | 3.80 ± 0.50 | -5.40 | < 0.001 e | 9.26 ± 0.24 | 4.43 ± 0.57 | -4.83 | < 0.001 e | -0.57 | 0.33 f |
| DHI. total | 59.44 ± 3.41 | 25.93 ± 2.79 | -32.93 | < 0.001 e | 60.62 ± 2.54 | 24.67 ± 3.2 | -36.53 | < 0.001 e | 3.60 | 0.40 f |
| DHI. emotional | 50.49 ± 3.41 | 22.5 ± 2.98 | -27.83 | < 0.001 e | 50.34 ± 3.09 | 19.7 ± 3.08 | -30.83 | < 0.001 e | 3.00 | 0.50 f |
| DHI. funtional | 67.93 ± 20.16 | 30.03 ± 21.2 | -37.90 | < 0.001 e | 72.93 ± 3.38 | 30.02 ± 4.4 | -42.92 | < 0.001 e | 5.02 | 0.37 f |
| DHI. physical | 61.39 ± 4.44 | 26.85 ± 2.83 | -34.53 | < 0.001 e | 62.41 ± 2.14 | 25.03 ± 2.98 | -37.38 | < 0.001 e | 2.85 | 0.54 f |
| THI. total | 48.14 ± 4.96 | 40.47 ± 4.5 | -7.53 | 0.023 e | 53.19 ± 5.04 | 48.19 ± 6.04 | -5.00 | 0.246 e | -2.53 | 0.81 f |
| THI. emotional | 44.39 ± 5.27 | 34.81 ± 4.37 | -9.58 | < 0.001 e | 53.98 ± 4.93 | 44.18 ± 5.19 | -9.80 | 0.002 e | 0.21 | 0.98 f |
| THI. functional | 51.22 ± 5.21 | 44.7 ± 4.82 | -6.53 | 0.070 e | 53.93 ± 5.38 | 49.54 ± 4.94 | -4.38 | 0.304 e | -2.15 | 0.78 f |
| THI. catastrophic | 45.90 ± 5.52 | 39.24 ± 5.31 | -6.67 | 0.147 e | 51.09 ± 5.85 | 52.26 ± 8.16 | 1.17 | 0.856 e | -7.83 | 0.39 f |
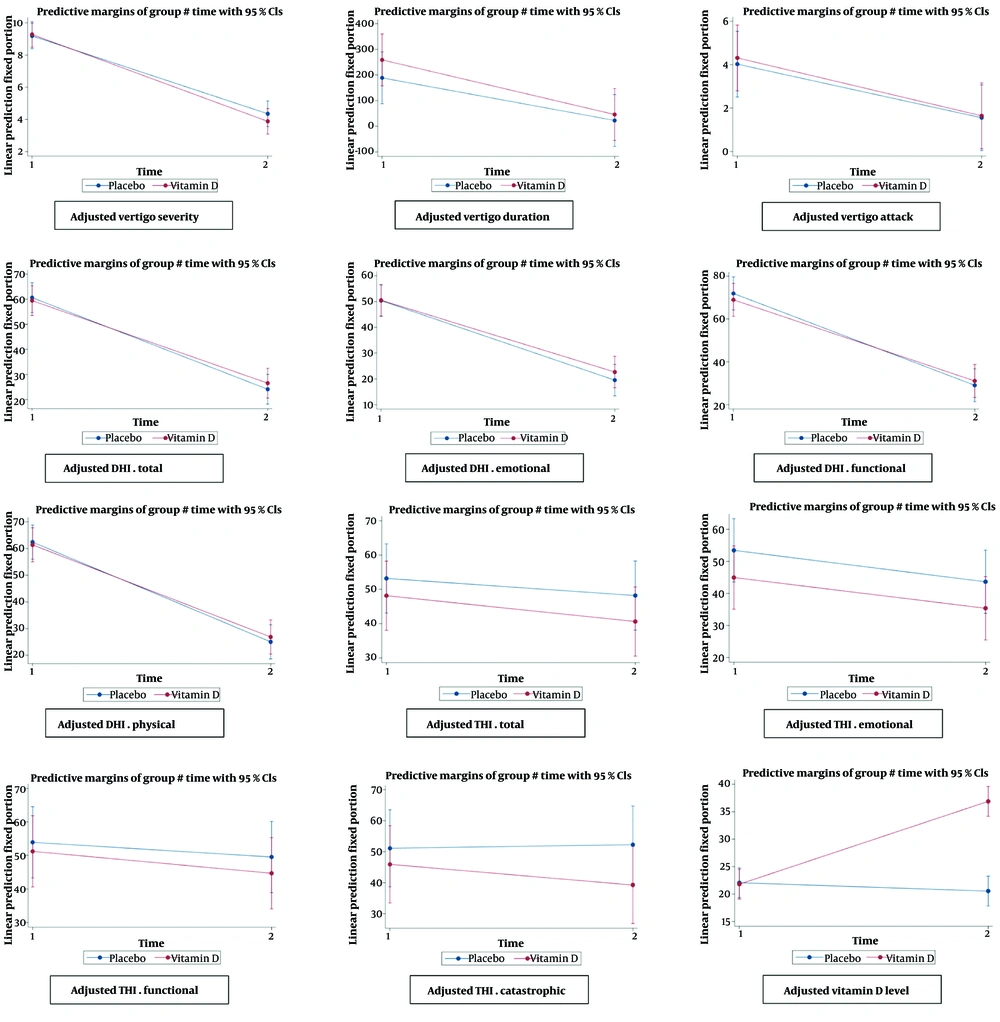
These findings suggest that while vitamin D supplementation might contribute to symptom relief, additional factors may influence the outcomes. As shown in Figure 2, there was a non-significant decreasing trend in the values of changes in THI-total, DHI-total, THI-functional, THI-emotional, DHI-functional, DHI-emotional, DHI-physical, vertigo duration, vertigo severity, and vertigo attacks in both study groups after adjusting for probable confounders. Mixed-effect regression models are shown in Appendix 1 in Supplementary File.
No serious adverse effects were reported during the trial. However, a few participants in the intervention group experienced mild gastrointestinal discomfort, such as nausea and bloating, which resolved spontaneously. No cases of hypercalcemia or other clinically significant side effects were observed.
5. Discussion
A limited number of investigations have been conducted on Meniere’s disease and its symptoms. The present clinical trial aimed to evaluate the effects of vitamin D supplementation on vertigo and tinnitus, the main complications in patients with Meniere’s disease. According to the findings from this trial, weekly supplementation with 50,000 IU of vitamin D for eight weeks did not significantly improve the number of vertigo attacks, the duration and severity of vertigo, or various assessed indices of dizziness and tinnitus.
However, within-group comparisons demonstrated a significant decrease in the mean number of vertigo attacks, the duration and severity of vertigo, various assessed indices of dizziness, and the score of emotional THI in both study groups at the final assessment, which might suggest a placebo effect. The notable improvements observed in both groups indicate the potential influence of a placebo effect. Factors such as patient expectations, frequent clinical visits, and increased attention from healthcare providers may have contributed to symptom relief regardless of the intervention.
Future studies should consider incorporating an additional placebo-controlled arm or objective biomarkers to better differentiate between the true effects of vitamin D supplementation and the placebo response. According to the proposed mechanisms of action of vitamin D and its beneficial impact on ear diseases, including Meniere’s disease, the lack of significant effects of vitamin D compared to placebo might be due to reporting bias caused by self-reporting of vertigo, tinnitus, and dizziness status by study patients.
Another possible explanation for the lack of significant findings is that the dosage and duration of vitamin D supplementation used in this study (50,000 IU per week for eight weeks) may not have been optimal for eliciting a therapeutic effect in Meniere’s disease. Moreover, variations in baseline vitamin D levels among participants could have influenced the response to supplementation, as individuals with adequate or near-adequate vitamin D status may not derive the same benefits as those with severe deficiency.
Future clinical trials should consider stratifying participants based on their baseline vitamin D levels and investigating different dosages and intervention durations to determine the most effective supplementation strategy.
The findings of studies by Sanchez et al. (24), Jeong et al. (25), Sheikhzadeh et al. (26), and Talaat et al. (27) are in disagreement with our results. Sanchez et al. (24) examined the effects of correcting vitamin D deficiency in patients with benign paroxysmal positional vertigo (BPPV), a temporal bone disease. They found that patients supplemented with vitamin D had a lower DHI score compared to the control group, and the probability of vertigo recurrence decreased after vitamin D administration. Additionally, a recent multicenter randomized clinical trial by Jeong et al. (25) demonstrated that co-supplementation of calcium and vitamin D reduced the occurrence of vertigo attacks in patients with confirmed BPPV. In another clinical study, Sheikhzadeh et al. (26) indicated that correcting vitamin D deficiency provides additional benefits to rehabilitation therapy in BPPV patients. Furthermore, a clinical investigation by Talaat et al. (27) reported a significant decrease in the recurrence rate of BPPV following therapy for severe vitamin D deficiency.
It should be noted that although BPPV and Meniere’s disease are both associated with vertigo, they differ completely in terms of pathophysiology. The pathophysiology of Meniere’s disease involves distension of the membranous labyrinth by endolymph, also known as endolymphatic hydrops (28). In contrast, the common pathophysiology of BPPV is the detachment of calcium carbonate otoconia from the macula of the utricle (29).
Moreover, Buki et al. (14) reported unpublished observations in their literature review regarding the effects of vitamin D supplementation in Meniere’s disease. According to their report, correcting vitamin D deficiency led to a substantial decline in the necessity for intratympanic gentamicin therapy.
The discrepancies between our results and the aforementioned studies may result from differences in the nationality, physiological status, lifestyle, and age group of study participants. The contradictory outcomes may also be attributed to the diverse methodological frameworks employed in the investigations, as well as the varying quantities and durations of vitamin D administration.
Given that the primary cause of Meniere’s disease is an initial viral infection followed by an autoimmune/auto-inflammatory reaction, vitamin D might alleviate these post-viral reactions by modulating the immune system response, suppressing inflammation via attenuation of nuclear factor kappa B (NF-kB) and mitogen-activated protein kinase (MAPK) pathways, stabilizing inner ear endothelial cells through its antioxidant potential, and promoting remyelination of the vestibular nerve (16, 30-33).
5.1. Strong Points and Weaknesses of the Trial
The present clinical trial exhibits several strengths, including strict eligibility criteria, acceptable adherence to the prescribed intervention, robust statistical analysis, and high racial homogeneity of the study population. However, certain limitations must be considered when interpreting our conclusions. First, the duration of supplementation may not have been sufficient to influence study outcomes. Second, potential confounding variables, such as physical activity and dietary intakes of the study patients, which could affect outcomes, were not assessed during the trial. Third, it is challenging to observe quantitative differences when vitamin D is used in combination with a vestibular suppressant (betahistine). Fourth, the sample size may be insufficient due to the consideration of an inappropriate effect size. Fifth, the generalizability of the results is limited to patients with Meniere’s disease affected by vitamin D deficiency, rather than all patients with Meniere’s disease. Finally, the self-reporting of study patients regarding their vertigo and tinnitus status might introduce reporting bias.
5.2. Conclusions
In summary, weekly supplementation with 50,000 IU of vitamin D for eight weeks did not significantly improve vertigo, dizziness, or tinnitus in patients with Meniere’s disease. Considering the proposed mechanisms of action of vitamin D and its beneficial effects, our results might be affected by some limitations. Therefore, more clinical trials with optimized methodological designs, including larger sample sizes and longer intervention periods, are required to investigate the efficacy of vitamin D in the treatment of Meniere's disease.